Juan Carlos Gómez Martín
Científico Titular
Consejo Superior de Investigaciones Científicas (CSIC)
Instituto de Astrofísica de Andalucia (IAA)


The troposphere is the lowermost layer of the Earth's atmosphere, where most meteorological phenomena occur. It extends to about 17 km at the equator and to about 7 km at the poles. Usually the troposphere is divided into the boundary layer (lowermost layer), the Free Troposphere and the Upper Troposphere-Lower Stratosphere (UTLS) region. Atmospheric chemistry is embedded in the complicated dynamics of the atmosphere. Seinfeld and Pandis, in the preface of their Atmospheric Chemistry and Physics book (Wiley, 1998), state that he atmospheric role of trace chemicals is disproportionate to their atmospheric abundance: they are responsible of phenomena ranging from urban pollution, acid deposition, stratospheric ozone depletion, to climate change. This is exactly the reason why halogens (fluorine, chlorine, bromine and iodine) have attracted so much scientific interest over decades. The 1995 Nobel prize went to Crutzen, Molina and Rowland for their work in atmospheric chemistry, particularly concerning the formation and decomposition of ozone. The 2016 Alfred Wegener Medal was awarded to Professor John P. Burrows for developing, adopting and applying instrumentation for remote sensing of the atmosphere, and initiating major advances in understanding the chemistry and dynamics of the atmosphere. Pioneering work by Chameides and Davis (1980) and Solomon et al. (1994) showed that both organic and inorganic iodine compounds -emitted from the sea surface and from marine biota- photo-dissociate rapidly in the troposphere to release iodine atoms, which then react with ozone to generate iodine monoxide (IO). A steady state is then established between I and IO as a result of the fast photolysis of IO, and therefore the two species are termed collectively as reactive iodine, or IOx = I + IO. The IOx react further with other species to generate different forms of inorganic iodine. The atmospheric chemical processing of iodine species influences the oxidizing capacity of the troposphere through catalytic ozone-depleting cycles and changes to the HOx (i.e. [HO2] / [OH]) and NOx (i.e.[NO2] / [NO]) ratios. Such chemical processing of iodine leads to its uptake and accumulation in aerosol, and subsequent transport and deposition on land, where it enters the continental ecosystems. Moreover, the clustering of iodine oxides triggers new particle formation events in the coastal boundary layer. Ozone depletion and iodine oxide clustering are to some extent competing processes: significant light-induced break-up of large, particle-forming iodine oxide molecules (IxOy) may result in regeneration of smaller, ozone-depleting iodine molecules IOx. Iodine chemistry may be responsible of up to 16% of the contemporary ozone removal in the tropical troposphere, making a negative contribution to the radiative flux. Uncertainties in these photochemical processes are particularly relevant, as anthropogenic ozone pollution has caused the global iodine source to increase by a factor of 3 since 1950. Our recent modelling work shows that short lived halogen compounds, whose oceanic emissions have been amplified by pollution, exert a significant indirect cooling effect on climate that needs to be considered in future IPCC assessments. I worked at the laboratories of John P. Burrows, John M. C. Plane and Alfonso Saiz-Lopez, from which I contributed to the understanding of atmospherically relevant chemical and photochemical reactions of iodine-containing molecules, particularly iodine oxides. I also built a resonance fluorescence instrument (ROFLEX) for detecting iodine atoms and molecules and coordinated the CHARLEX field campaign in the Equatorial Pacific led by Alfonso Saiz-Lopez. Recently, I have led experimental studies conducted at CSIC and the University of Leeds on the photochemistry of IxOy, the role of iodine oxydes and oxyacids in new particle formation and the reactions that may interfere in CIMS detection in the field of these species.
Iodine oxides are ubiquitous in the marine and polar boundary layers, where they form by ozone-driven photo-oxidation of iodocarbons and molecular iodine emitted from the sea or ice as a result of both biotic and abiotic processes. From early laboratory studies on gas-phase iodine chemistry it is well known that the recombination of iodine monoxide (IO) and dioxide (OIO) precedes the formation of condensable chemical species, which can undergo nucleation and lead to the formation of optically detectable aerosol and solid deposition. In fact, it has been shown that emission fluxes of molecular iodine from coastal macroalgae under oxidative stress at low tide are strong enough to sustain substantial secondary aerosol formation in the atmosphere. This potential link between marine biota and climatically sensitive variables such as condensation nuclei and ozone has motivated numerous laboratory field studies in recent years. I have made significant contributions to the understanding of the mechanism of iodine oxide catalyzed ozone destruction and particle formation. My PhD thesis with John P. Burrows was entitled ‘spectroscopic, kinetic and mechanistic studies of atmospherically relevant I2/O3 photochemistry’. In the course of my PhD work, I determined the visible absorption cross sections of IO and OIO and the rate constant of the IO self-reaction and the branching ratios of its product channels. I hypothesized that the IO+OIO and OIO+OIO recombination reactions would be the path to the formation of iodine oxide particles, as I demonstrated later at John Plane’s lab. In 2009, my photofragment excitation experiments with three lasers and an iodine microwave discharge lamp solved the long-standing issue of the photolysis of OIO above 500 nm. we found that OIO photodissociation yields, surprisingly, iodine atoms with unit quantum yield. In 2013 I reported the first direct experimental evidence of the I2O2, I2O3, I2O4, I2O5 and I3O7 molecules in the gas phase by using photo-ionization time-of-flight mass spectrometry. Recent field and chamber research using Chemical Ionization Mass Spectrometry (CIMS) suggest a role of iodic acid (HOIO2) in coastal new particle formation, but the atmospheric source of HOIO2 and its importance in new particle formation compared to IxOy are uncertain. In collaboration with the University of Leeds, we have recently determined the absorption cross sections of various IxOy molecules in the actinic spectral window by combining experimental photodepletion experiments at 355 nm and 532 nm with ab initio calculations. We have also carried out experiments to investigate possible sources of HOIO2 and to elucidate the mechanism of iodine oxide particle formation. In a paper published in JACS we have investigated the ion-neutral chemistry that takes place in the nitrate CIMS inlet when iodine oxides and oxoacids are sampled and we have discussed the mechanism of HOIO2 formation in the atmosphere and the interpretation of CIMS spectra. We have also carried out experimental and theoretical work on iodine chemistry on air-aerosol interfaces. All these laboratory results are being implemented in collaboration with Alfonso Saiz-Lopez in a global chemistry climate model (CAMCHEM) to evaluate the impact of iodine chemistry on ozone depletion through the troposphere. In the near future, a iodine new particle formation mechanism will be added to the model in order to evaluate the global importance of this source of new atmospheric particles. Although my focus has swung towards atmospheric aerosol and cosmic dust, I have contributed laboratory and theoretical work over the last five years to the ERC-CoG project ‘Quantifying the climate dimension of natural halogens in the Earth system: Past, present, future’ (CLIMAHAL) and I remain active in this field.
The detection of gas phase atomic species in the atmosphere requires in situ techniques with an excellent detection limit and a compact, portable and robust set-up. The techniques based on atomic resonance fluorescence excited by microwave or radiofrequency discharge lamps offer all these advantages, as compared e.g. to the more sensitive but heavier, more complex laser induced fluorescence approaches. Discharge lamps have been utilized for the detection of stratospheric halogen species over 3 decades by research groups at Harvard and Jülich. However, in 2009 we realized that the potential of the lamp based techniques in the boundary layer had been only partially explored, as a result of the technical challenges posed by collisional quenching, background signal and statistical noise caused by scattered light and the requirement of an accurate calibration of the fluorescence signal. At the Laboratory for Atmospheric and Climate Science of the Spanish Research Council (CSIC) in Toledo, we initiated the construction of a new line of machines based on atomic resonance fluorescence detection of halogen atoms. The first prototype dedicated to the detection of atomic iodine was built in 2010. Our concept adds the capability of measuring simultaneously off-resonance fluorescence from molecular iodine, I2. The system is called Resonance and Off-Resonance Fluorescence by Lamp EXcitation (ROFLEX). The core of the instrument is an expansion chamber (< 50 Torr) where atoms and molecules are excited by a radiofrequency discharge lamp built at the IAPS Latvia and florescence is collected at right angles by sensitive photon counting detectors (Perkin Elmer). Automation and housekeeping are provided by an IGI-Systems interface. Calibration of the molecular signal is achieved by using Cavity Enhanced Absorption Spectroscopy, whereas the atomic signal is calibrated by the photolysis of known amounts of molecular iodine. Thee system was deployed in the LEGOLAS and CHARLEX campaigns. The laboratory for Atmospheric and Climate Science of the Spanish Research Council (CSIC), initially in Toledo is now the AC2 department of the Institute of Physical Chemistry Blas Cabrera in Madrid. Currently I collaborate with AC2 leader Alfonso Saiz-Lopez in an upgrade to the ROFLEX machine for future field campaign deployment. The ROFLEX has also been used to carry out laboratory experiments on atomic iodine reactivity.
The Climate and HAlogen Reactivity tropicaL EXperiment Chemmistry (CHARLEX) was conducted in the Galápagos Islands between 2010 and 2012. It was led by the Laboratory of Atmosphere and Climate Science of CSIC (currently AC2 department of the Institute for Physical Chemistry Blas Cabrera), with Alfonso Saiz-Lopez as PI. There is a permanent meteorological station at San Cristobal Island from which ozonesondes of the SHADOZ program have been launched over more than a decade (at the peak of the SHADOZ program the maximum launch frequency was once a week). However, CHARLEX was the first long term atmospheric chemistry and composition field campaign in the Galápagos Islands, with ozone, NOx, mercury, aerosol, halocarbons and trace gases being measured on a daily basis with high sampling frequency. The project was certified by the Galápagos National Park and was logistically supported by the Ecuadorian INHAMI and the Charles Darwin Foundation. Anoop Mahajan, Tim Hay and myself were the backbone of the campaign, spending six months each at the remote Isabela Island over a period of 2 years. Scientist of other collaborating institutions that spent some time at Galápagos with their instruments were: Samantha MacDonald and John Plane (University of Leeds), Feiyue Wang, Debbie Armstrong and Marcus Lemes (University of Manitoba), Maria Concepcion Parrondo and Mar Sorribas (INTA). CHARLEX was motivated by high concentrations of active halogens measured at Cape Verde and the apparent positive detection of iodine monoxide (IO) above the equatorial Eastern Pacific by ENVISAT/SCIAMACHY. Iodine oxides are known to be very efficient at catalytically destroying ozone and at trigger new particle formation in coastal areas. The hypothesis of the campaign was that high halogen precursor emissions from a high productivity part of the ocean would result in high IO concentrations in the boundary layer, which in turn would destroy surface ozone and drive new particle formation in an remote, ultraclean environment. This was a strong motivation since it could imply a strong chemistry-climate interaction. Another hypothesis was an important role of halogen in anthropogenic mercury oxidation. The campaign demonstrated the presence of IO but at low concentrations. This would be responsible of a sizeable portion of the ozone destruction over the ocean, but would be not able of sustain significant particle formation against scavenging by background sea-salt aerosol. Nevertheless, the campaign demonstrated, together with the HaloCAST and Malaspina Cruises, that iodine oxides are ubiquitous over the global oceans. It also documented the mercury cycle, the particle environment and the ozone-NOx variability. Evidence of coupling between intraseasonal variability of ozone and sea surface temperature (the pacific tropical instability wave) was observed. Subsequent lab studies at York and Leeds led us to the conclusion that ozone deposition and reaction with iodate ions in the sea-air interface is a temperature dependent global source of iodine to the atmosphere that needs to be included in the model inventories for assessing the impact of iodine on tropospheric ozone.
References:Iodine is a key trace element in continental food chains whose major global source is oceanic surface gas emissions of iodine-bearing molecules to the atmosphere. Atmospheric chemical processing of these substances is followed by incorporation of the iodine-bearing products into airborne marine aerosol particles, which are the carriers of iodine to the continents. We have compiled a data set of aerosol composition measurements reporting iodine concentrations at many different locations, seasons, and years. Analysis of the variation of the concentration iodine in aerosol with latitude and longitude enables us to confirm the main mechanism emitting iodine from the oceans, which is triggered by deposition of ozone on the water-air interface. We have also analyzed the seasonal variation of the iodine concentration in aerosol in different locations, which also sheds light onto additional iodine sources to the atmosphere. Unfortunatelly, long-term trends could not be stablished unsing these observations, although they are compatible with the model-predicted enhancement of aerosol iodine as a result of increased oceanic iodine emissions over the last 50 years related to ozone pollution.
In collaboration with IQFR-CSIC and researchers at the University of East Anglia, we are tring to shed light onto the chemical mechanism governing the speciation of iodine in marine aerosol. For this, we have also compilled a global data set of iodine speciation measurements. This mechanism, which appears to be driven by acidity and availability of dissolved organic matter, determines under which circumstances aerosol is a sink for iodine or a reservoir that recycles gas-phase iodine.
References:Atmospheric aerosols play key roles in climate and have important impacts on human activities and health. Hence, much scientific effort has been directed towards developing methods of improved detection and discrimination of different types of aerosols. Among these, light scattering-based detection of aerosol offers several advantages including applications in both in situ and remote sensing devices, bulk detection and multi-angle and multi-wavelength measurements. At the Cosmic Dust Laboratory (CODULAB) of IAA-CSIC, we are carrying out scattering matrix measurements of samples of airborne desert dust, volcanic ashes, bioaerosols and other major types of atmospheric aerosol. Our aim is providing field researchers with criteria and reference data that can be used to detect and indetify different types of aerosol in atmospheric observations. The scattering matrix data can be donwloaded from the Granada-Amsterdam Light Scattering Database.
References: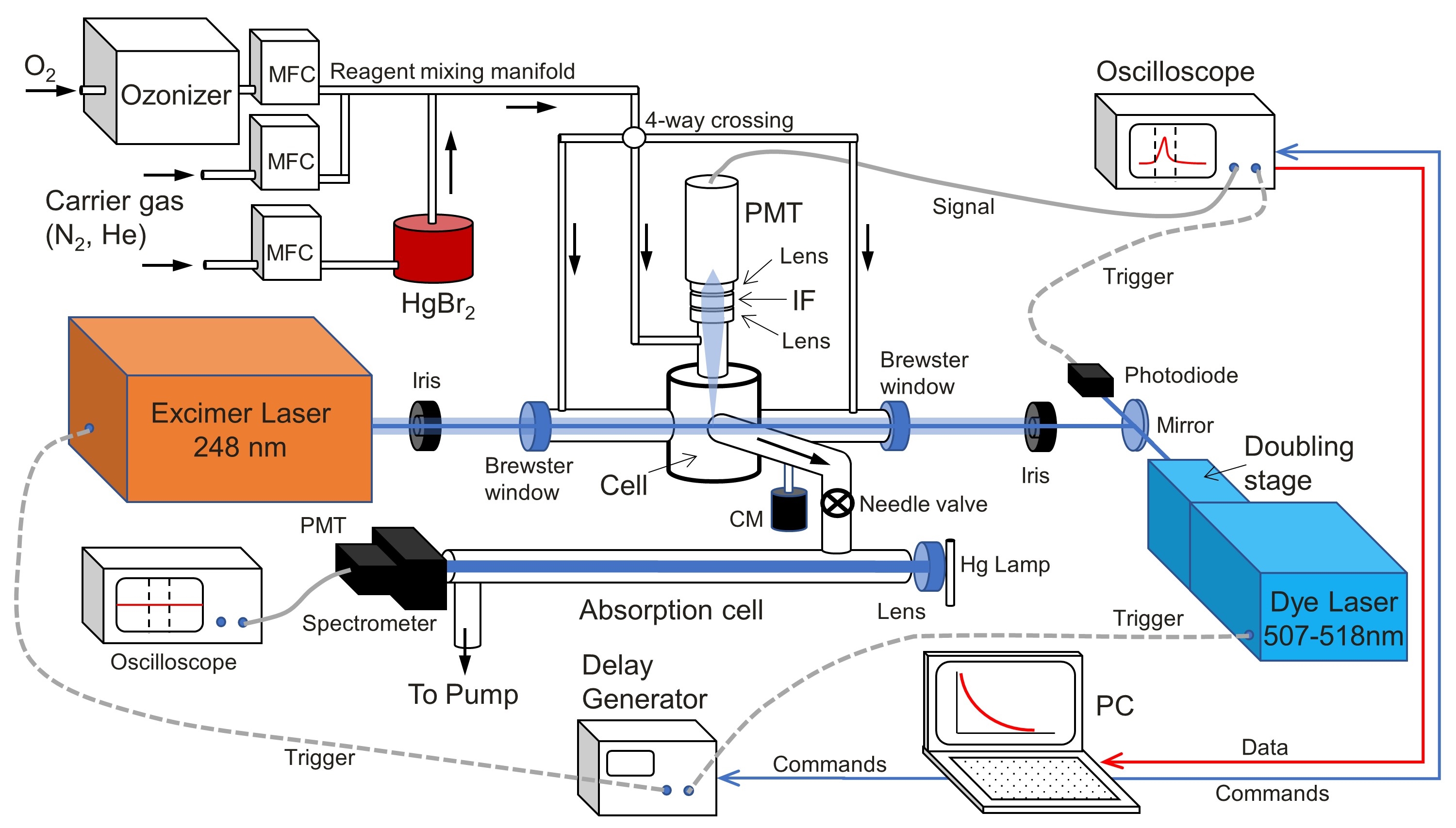
Mercury is a neurotoxic pollutant emitted mainly in its elemental form Hg0 to the atmosphere where it is oxidized to reactive HgII compounds. The atmosphere is a key component of the global Hg cycle because it facilitates its chemical processing and planetwide dispersion. The first oxidation step is initiated by photochemically produced Br atoms to form HgBr, a radical HgI intermediate that can then photolytically or thermally decompose back to Hg0, or be further oxidized to HgII compounds by atmospheric radicals (Saiz-Lopez et al. 2020). Very few of these reactions have been studied experimentally in the laboratory. Recently, we carried out measurements of the rate constant of the reaction between HgBr and ozone with the classical Pulsed Laser Photolysis-Laser Induced Fluorescence set-up at the University of Leeds in collaboration with Prof. John Plane. We are planning to continue with these studies in the new kinetics and particle formation laboratory (LABSPARKS) that I am leading at IAA-CSIC.
References: